Malaria Transmission Biology
 Our research has led to the discovery of a mosquito-based malaria transmission-blocking vaccine (TBV) candidate (the AnAPN1 TBV), which has recently completed process development studies and is under consideration for First-In-Human trials in the near future. We were the first to provide proof of concept demonstrations of the potential utility of mosquito based TBVs to control both human malaria parasite species (P. falciparum and P. vivax) in diverse anopheline vector species across the globe. Our multi-OMICS strategy perspective has also led to the discovery of a novel small molecule with potent malaria transmission-blocking properties, the identification of the Plasmodium protein target that is blocked by this small molecule, the completion of the P. falciparum life cycle in vitro (liver stages), a novel saliva-based rapid diagnostic test for malaria, and new insight into vector host-parasite interactions during oocyst development, to name a few. To support the discovery and translational research pipeline, we are also developing novel, single dose drug and vaccine delivery methods that are appropriate for malaria endemic countries in the developing world. TEAM MALARIA is composed of 4 postdoctoral associates, 1 PhD student, the lab manager, Center Program Coordinator, and a lateral flow chemist. One postdoc and PhD student also serves as a bridge with TEAM ARBO.
Our research has led to the discovery of a mosquito-based malaria transmission-blocking vaccine (TBV) candidate (the AnAPN1 TBV), which has recently completed process development studies and is under consideration for First-In-Human trials in the near future. We were the first to provide proof of concept demonstrations of the potential utility of mosquito based TBVs to control both human malaria parasite species (P. falciparum and P. vivax) in diverse anopheline vector species across the globe. Our multi-OMICS strategy perspective has also led to the discovery of a novel small molecule with potent malaria transmission-blocking properties, the identification of the Plasmodium protein target that is blocked by this small molecule, the completion of the P. falciparum life cycle in vitro (liver stages), a novel saliva-based rapid diagnostic test for malaria, and new insight into vector host-parasite interactions during oocyst development, to name a few. To support the discovery and translational research pipeline, we are also developing novel, single dose drug and vaccine delivery methods that are appropriate for malaria endemic countries in the developing world. TEAM MALARIA is composed of 4 postdoctoral associates, 1 PhD student, the lab manager, Center Program Coordinator, and a lateral flow chemist. One postdoc and PhD student also serves as a bridge with TEAM ARBO.
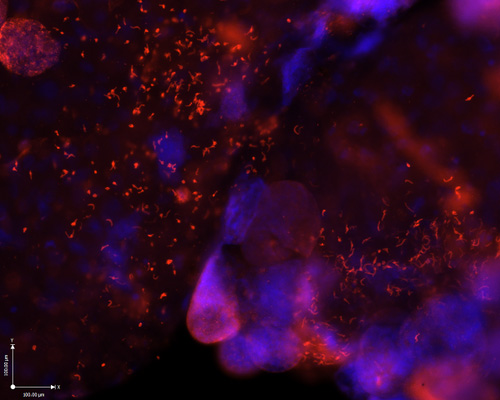
[Pictured: Anopheles gambiae midgut infected with P. falciparum oocysts that have burst open to release thousands of sporozoites]
Contact Us
Learn more about the research in the Dinglasan Laboratory.
Latest News
STAY UP TO DATE WITH OUR LATEST NEWS. LEARN ABOUT NEW DEVELOPMENTS, TREATMENTS, VACCINES, OUR LAB & MUCH MORE!

UF Malaria Researcher and Global Partners Win NIH Award for New International Center of Excellence
- Posted OnApril 24, 2024
- By epiadmin
The cooperative agreement, from the National Institute of Allergy and Infectious Diseases, will establish a new International Center of Excellence in Malaria Research (ICEMR) focused
Read More
UF Professor of Infectious Diseases, Dr. Rhoel Dinglasan, discusses the new saliva COVID tests
- Posted OnAugust 20, 2021
- By epiadmin
Watch WCJB TV20 video here.
Read More
First in human clinical trials of the AnAPN1 malaria transmission-blocking vaccine
- Posted OnMarch 31, 2021
- By epiadmin
Former director of the PATH Malaria Vaccine Initiative, Christian Loucq, M.D., says it is exciting to see Dinglasan’s concept moving toward a clinical trial. “There
Read More